Eye Drop Recall 2023
- 550+ Google 5-Star Reviews
- $1 Billion won over 15 years
- Won’t settle for low offers, not afraid to fight for max value
Get a Free Case Evaluation
No fees until we win!
All San Diego Case Types

Personal Injury

Brain Injury

Car Accident

Slip & Fall

Truck Accident

Wrongful Death
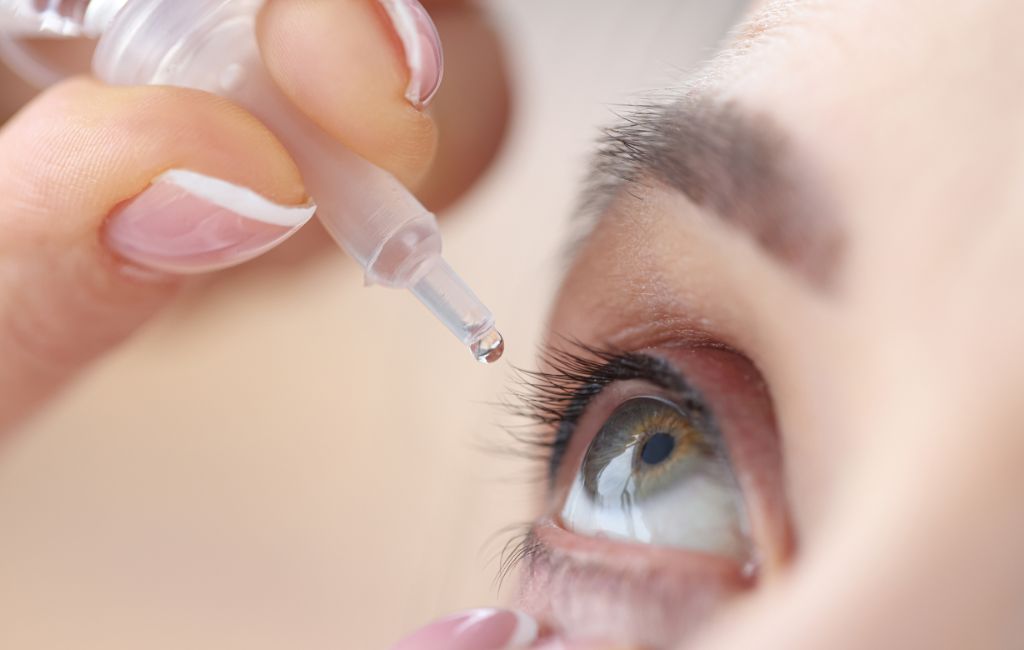
Eye Drop Recall
The Food and Drug Administration (FDA) is investigating multistate outbreak related to dangerous eye drops that have caused vision loss and even several deaths. As a result of the FDA’s investigation—in conjunction with the Center for Disease Control (CDC) and several state agencies—three companies have initiated recalls of their injury-causing products.
It’s important to pay attention to eye drop recalls, as they could spell danger for those who take them. A recent string of recalls of eye drops has put many people on edge. People who have purchased the recalled product may be left wondering what this means for their health and whether they can continue using the product or not. In this post, we will explore what happened with the recent recall, which types of eye drops were affected, and how you can protect yourself from future recalls like these. With all that in mind, let’s dive into the world of eye drop recalls!
What are the eye drop recalls and why were they used?
FDA recalls regarding several brands of eye drops have been making headlines lately and have left many people wondering what exactly is going on. Essentially, the recalls were issued due to concerns over contaminated eye drops. Specifically, the recalls affect certain over-the-counter eye drops and ointments manufactured by Global Pharma Healthcare, Pharmedica, and Apotex. Following an investigation by the Centers for Disease Control and Prevention (CDC) into a series of bacterial infections that spanned multiple states and were suspected to be linked to the use of tear drops, a recall was initiated. The recall was prompted by 68 reported incidents of negative reactions to the drops, such as eye infections, irreversible vision impairment, and even three deaths resulting from a bloodstream infection.
What brands of eye drops have been recalled?
Global Pharma Healthcare issued a recall on February 2, 2023, for its EzriCare and Delsam Pharma brands due to potential bacterial contamination. The recall was initiated following an investigation by the Centers for Disease Control and Prevention (CDC) into a series of bacterial infections that spanned multiple states and were suspected to be linked to the use of the eye drops. Global Pharma Healthcare warned consumers that using the contaminated eye drops could lead to blindness and advised them to immediately stop using the drops.
Additionally, on February 24, Global Pharma issued another recall for one batch of Delsam Pharma’s “Artificial Eye Ointment” due to possible microbial contamination. Although using the contaminated ointment could cause infections leading to blindness, the company had not received any reports of injuries related to the product.
On March 1, Apotex also issued a recall for several lots of its own brand of glaucoma drops, called “Brimonidine Tartrate Ophthalmic Solution, 0.15%,” due to concerns that cracks in some of the units’ caps could compromise the drops’ sterility and lead to infection. The company has observed four bottles with cracked caps, but there have been no reports of negative health outcomes related to the product. Apotex initiated the recall “out of an abundance of caution.”
Finally, on March 3, Pharmedica USA recalled two lots of “Purely Soothing, 15% MSM Drops” over sterility concerns. The pharmaceutical company warned that using the contaminated eye drops could lead to blindness, but it had not received any reports of infection or illness related to its product.
What should you do if you’ve used these drops?
If you’ve used these drops, it’s important to understand the potential effects and what steps you should take. These eye drops have caused blurry vision, eye pain and discomfort, redness and irritation, vision loss, and in some cases, deaths have been reported. The first thing you should do if you’ve used any of these brands or products is call Gomez Trial Attorneys for a free, no obligation case evaluation. Their team of experienced lawyers will work with you to see if you have been injured because of these recalled eye drops. If you have had any reported issues with these drops, it’s imperative that you call immediately.
What We Got
$2,800,000
Original Offer
$481,000
Our Process... Easy as 1. 2. 3!
Call Us
We will determine your case and submit
We get to work
You will get regular update from us
Win
Collect your compensation
-
“John helped me find doctors, he referred me to his neurologist, his physical therapist, I mean, anything I needed he was right there, every step of the way. I couldn’t have asked for a better result from all of this, I would absolutely recommend Gomez Trial Attorneys.”
-
“During the time I was working with Gomez Trial Attorneys, they treated me very, very well. 100% of the time, they believed me, and they were very compassionate. They felt sorry for what happened and they understood the therapy process.”
-
“They held my hand the whole time and kept me in the loop every aspect of my case which was very refreshing to me. They helped me get my settlement offer as fast as possible and I was able to keep my farm”
-
“The Gomez experience was the best experience it could be for me really, only positive things to say. They really were there every step if the way. Thanks to Gomez Trial Attorneys my dad is able to support my family as a single father”
-
“He opened the door for me to join his firm to help other brain Injury survivors and I never met another firm who is like this who was so understanding and caring who took the extra step and walked the extra mile with their clients and this is the best”
-
“I am very satisfied with the outcome with Gomez and I would definitely recommend Gomez to anybody, we tell people all the time, Get Gomez! They are really thorough with everything and they make you feel real comfortable.”
-
“Just helped us through, guided us through, I kept notes all those years, we had questions all the time and they would always keep us informed of what was going on. They just unlayered it, layer by layer, I’ve never seen anything like them. Thank God for them.”


